New IlluminOss Bone Stabilization System for Treatment of Traumatic and Fragility Fractures of The Humerus, Radius and Ulna
Press Release Summary:
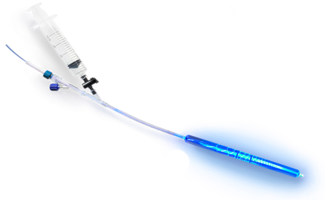
- Enable surgeons to create a conforming implant for each individual patient
- FDA registered system have visible blue light that rapidly cures the implant immediately stabilizing the fracture
- Utilizes expandable balloon implant delivered in minimally invasive manner through a small incision to support poor quality, compromised bone
Original Press Release:
IlluminOss Medical Launches The IlluminOss Bone Stabilization System for Use in The Treatment of Traumatic and Fragility Fractures in The U.S.
The Company’s Innovative Fracture Repair Technology Provides a New and Compelling Solution for Fragility Fractures Associated with the Aging Population
East Providence, RI (Feb 26, 2019) – IlluminOss Medical, Inc., a privately held, commercial-stage medical device company focused on minimally invasive orthopedic fracture repair, today announced the U.S. launch and commercial availability of the IlluminOss Bone Stabilization System for use in skeletally mature patients in the treatment of traumatic and fragility fractures of the humerus, radius and ulna. IlluminOss recently obtained FDA clearance for these exciting new indications*, having been previously available solely for the treatment of pathological and impending pathological fractures associated with metastatic disease in the U.S. IlluminOss Medical, Inc. will be in Booth #6243 at the AAOS 2019 Annual Meeting in Las Vegas, NV from March 12-16th and will be presenting data regarding the IlluminOss System on March 13th at 1:45 pm in Exhibit Hall B at Booth #3032.
“We are thrilled to announce the commercial launch of the IlluminOss System,” said Jeff Bailey, CEO, IlluminOss Medical. “IlluminOss is truly an innovative addition to fragility fracture treatment and we have been receiving outstanding feedback from our orthopedic surgeon customers regarding its potential to make a difference for patients with poor quality bone. Entry into the multi-billion dollar U.S. trauma market marks a significant milestone for our company.”
The IlluminOss System utilizes an expandable balloon implant that is delivered in a minimally invasive manner through a small incision to support poor quality, compromised bone. When the implant is infused with a monomer, it conforms to the shape of the patient’s intramedullary bone canal. A visible blue light rapidly cures the implant immediately stabilizing the fracture. Surgeons utilizing IlluminOss have reported reduced hospital stays, shorter procedure times, less post-operative pain, less use of pain medications and patients returning to their daily living activities faster.
"IlluminOss provides an exciting new option in fracture repair for difficult-to-treat osteoporotic or other patients with compromised bone, and is a much-needed addition to conventional treatment with nails, plates and screws,” said Dr. Mark Goodman, University of Pittsburgh Medical Center. “The fact that this unique technology was designed specifically for use in patients with compromised bone enables surgeons to create a conforming implant for each individual patient and is a game-changer in fracture repair."
The IlluminOss System received its initial de novo FDA clearance in December 2017 for fractures associated with metastatic bone. Clearance for traumatic and fragility fractures was received in August 2018. The IlluminOss System has been commercially available in international markets and has been in clinical use since 2010.
About IlluminOss Medical
IlluminOss Medical is a privately held, commercial-stage medical device company focused on designing, developing and marketing orthopedic fracture repair products that leverage its proprietary bone stabilization technology, the IlluminOss System.
IlluminOss’ minimally invasive technology produces patient-specific intramedullary implants for fracture fixation by utilizing a light-curable polymer, contained within an expandable balloon catheter, to achieve bone stabilization. The revolutionary procedure uses a small percutaneous surgical approach, providing patients and clinical with a fast, patient-specific method of orthopedic bone stabilization.
The company currently markets its products in international countries under a CE Mark for approved clinical applications through both a direct sales force and distribution networks and has obtained U.S. Food and Drug Administration (FDA) clearance.
*The IlluminOss Photodynamic Bone Stabilization System (PBSS) is indicated for use in skeletally mature patients in the treatment of traumatic, fragility, pathological, and impending pathological fractures of the humerus, radius, and ulna. The IlluminOss Photodynamic Bone Stabilization System can also be used in conjunction with an FDA-cleared fracture fixation system to provide supplemental fixation in the humerus, radius, and ulna.
Contact:
Jordan Bouclin
SVM Public Relations
(401) 490-9700




