New BioQuantiPro CHO-HCP ELISA Kit is Designed with Validated Antibodies of Rockland
Press Release Summary:
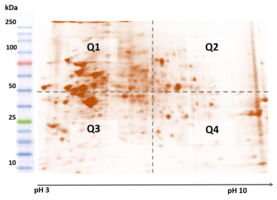
The BioQuantiPro™ CHO-HCP ELISA Screening Kit is used for detecting and screening of host cell protein contaminants in bioprocessing. The kit measures analytes from crude cellular harvests, cell culture lysates and process intermediates. It provides high coverage of generic CHO-HCP kits and low variability.
Original Press Release:
Rockland Announces General Release of BioQuantiProâ„¢ Host Cell Protein Screening Kit for Bioprocessing
Rockland Immunochemicals, Inc. announced the general release of their new BioQuantiPro™ CHO-HCP ELISA Kit for detection and screening of host cell protein (HCP) contaminants in bioprocessing.
LIMERICK, PA. (PRWEB) SEPTEMBER 04, 2018 - Rockland Immunochemicals, Inc. announced today the general release of their new BioQuantiPro™ CHO-HCP ELISA Kit for detection and screening of host cell protein (HCP) contaminants in bioprocessing. The kit was specifically designed with Rockland’s novel, validated antibodies to detect HCP contaminants and to measure analytes from crude cellular harvests, cell culture lysates, and process intermediates. The BioQuantiPro CHO-HCP ELISA Kit boasts the highest coverage of generic CHO-HCP kits, low variability, and, most importantly, the data to back it all up.
“While working with bioprocessing clients, we consistently hear the same complaint about HCP kits: ‘The generic kit I am using works OK, but I wish I had reliable data that I can trust for the validation of the antibodies used in the kit’” commented Karin Abarca-Heidemann, PhD., Vice President of Science Operations. “We aimed to create a kit that fills the void by providing transparency into the data necessary to develop accurate bioprocessing standards with confidence. We are proud of what we have accomplished with this new kit and look forward to working with customers to advance the science of bioprocessing.”
Rockland has a long history of developing process-specific HCP antibodies in the biotherapeutics field. “Over the last 15 years we have successfully developed and manufactured custom process-specific HCP antibodies for our customers. In the design and function of this CHO-HCP kit, we drew upon our experience gained from working with a diverse set of biopharmaceutical clients. We are eager to see the feedback of the scientists who need this kit,” said David Chimento, PhD., Vice President of Client Solutions.
The BioQuantiPro CHO-HCP ELISA Kit is the first of several bioprocessing kits Rockland expects to release over the next eighteen months. Expect more and get more from Rockland in forthcoming kits for HEK 293 and Escherichia coli expression systems and other common bioprocessing contaminant targets.
For more information on the BioQuantiPro CHO-HCP ELISA Kit, please visit https://rockland-inc.com/cho-hcp/.
Contact Information
Christina Rowley
Rockland Immunchemicals, Inc.
http://https://rockland-inc.com
484-791-3823




