Microbiologics Introduces New Molecular Diagnostics Panels with Multiplexed Pellets
Press Release Summary:
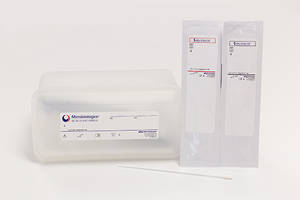
The Molecular Diagnostics QC Panels include Group A Streptococcus and respiratory control panels. The panels are designed for performing respiratory and Group A Streptococcus testing. They come with features like room temperature storage and swabs that mimic patient sample processing.
Original Press Release:
Microbiologics Expands Its Molecular Diagnostics QC Offering for Respiratory Assays
Two new control panels launched for respiratory and Group A Streptococcus testing
SAINT CLOUD, Minn., Dec. 13, 2018 /PRNewswire/ -- Microbiologics, Inc. has launched two new panels for quality control of Group A Streptococcus (GAS) and respiratory molecular assays and test methods to help clinical laboratories deliver accurate results during cold and flu season. The Group A Streptococcus (GAS) Control Panel and Respiratory (21 Targets) Control Panel include convenient features such as multiplexed pellets, swabs that mimic patient sample processing and room temperature storage.
"Consistency and ease-of-use are always top of mind for our team when we develop new products," said Brian Beck, Ph.D., Vice President of Research and Development. "Whether it's preserving pathogens on a collection device or multiplexing respiratory targets at concentrations that appropriately challenge an assay or test, we consider the sample workflow and engineer convenience and quality into all of our controls."
Microbiologics now offers a full spectrum of inactivated and live culture controls for clinical laboratories performing respiratory and GAS testing including:
- Group A Streptococcus (GAS) Control Panel (Catalog #8219) – Inactivated swab format
- Respiratory (21 Targets) Control Panel (Catalog #8217) – Inactivated pellet format
- Cepheid Xpert® Respiratory Control Panel (Catalog #8199) – Inactivated swab format
- Helix Elite™ Inactivated Standards (inactivated pellet format):
- Visit microbiologics.com to view all Helix Elite™ Inactivated Standards and live culture strains for respiratory and GAS testing.
"Our customers rely on us to provide controls they can trust, especially during flu season when clinical labs are required to deliver accurate results while maintaining fast turnaround times," said Brad Goskowicz, Chief Executive Officer. "Partnering with diagnostic companies to improve patient care by supporting rapid molecular testing will continue to be a top priority for our team."
A full list of the Microbiologics verification and quality control panels for common instruments including the BD MAX™ and Cepheid GeneXpert®, and syndromic assays including Blood Culture Identification (BCID), Women's Health/STI and Healthcare-Associated Infections (HAIs), can be found at www.microbiologics.com.
About Microbiologics: Microbiologics, Inc. provides the highest quality test-ready QC microorganisms and molecular controls for assay development, instrument validation and routine quality control testing of rapid diagnostic assays in the clinical, pharmaceutical, food, water and educational industries. Microbiologics produces over 1000 strains in a variety of user-friendly formats including qualitative, quantitative, Certified Reference Material and Parasite Suspensions. Microbiologics has an expansive international distribution network and ships to over 140 countries. The company is known for its exceptional customer service and knowledgeable technical support staff.
Laura Bebo-Ekanayake
Microbiologics
320-217-6606
lbebo@microbiologics.com




