Foster Corporation Launches Lazermed Laser Marking Technology that Offers High Resolution and Durable Printing Capability
Press Release Summary:
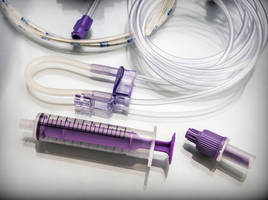
- Offers permanent, easily readable and eco-friendly alternative to solvent based printing technologies for medical device applications
- Weather durable, available for high temperature applications and have passed USP Class VI testing
- Used for injection molded, extruded, and blow molded components
Original Press Release:
Foster Corporation Introduces Lazermedâ„¢ Laser Marking Technology for Medical Device Applications
LazerMed™ laser marking technology offers a permanent, easily readable and eco-friendly alternative to solvent based printing technologies for medical device applications
Putnam, CT USA - (January 22, 2019) - Foster Corporation, a leader in polymer solutions for medical device and pharmaceutical applications, introduces LazerMed™, a biocompatible laser marking technology that offers high resolution, durable printing capability for plastic materials used in medical devices such as tubing, catheter shafts, labware, and medical equipment.
Laser marking is a contact-free process that provides for ultra-fast printing that is eco-friendly (no solvents), with no pre-treatment of substrates required. In addition, there are no consumables (inks) required, as the laser marking pigments are melt blended into the polymer at the compounding stage. This proprietary technology has no effect on the sterilization process and can be added to a polymer as a masterbatch. The result is a permanent solution that can be used for injection molded, extruded, and blow molded components.
The LazerMed technology is manufactured using state-of-the-art laser marking pigments. These pigments provide a versatile printing platform that can be used in the vast majority of plastic materials, including transparent and opaque materials, used in medical device applications. Laser marking pigments are weather durable, available for high temperature applications and have passed USP Class VI testing for biocompatibility.
“LazerMed laser marking technology benefits medical device manufacturers by offering an eco-friendly cost effective alternative to traditional printing technique,” said Tim Largier, Manager of Research & Development at Foster. “It benefits users at the delivery level by providing a permanent and safe method for labeling medical devices and components.”
For more information on Foster’s LazerMed laser marking technology, please visit Foster Corporation at Booth #2923 at MD&M West show on February 5-7, 2019 in Anaheim, CA or go to www.fostercomp.com.
Contact:
Kris Jommersbach
Gemini Communications
PH: +(1) 610-935-1633




