Single-Use Biocontainers feature end-ported design.
Press Release Summary:
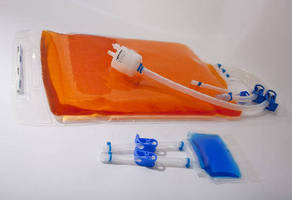
Designed to meet requirements of biopharmaceutical industry, TepoFlex® Biocontainers are made of polyethylene-based TepoFlex® film with combined gas and water vapor barrier that protects product stored within them. Units are available with silicone or TPE tubing with variety of connections, and can be specified with or without capsules. Certified animal component free, biocontainers come in 50, 250, and 500 mL sizes as well as 1, 2, 5, 10, and 20 L sizes.
Original Press Release:
End-ported Single-Use Biocontainer Standards - 50 mL to 20 L
Meissner now offers end-ported TepoFlex® biocontainer standards in sizes of 50 mL, 250 mL, 500 mL, 1 L, 2 L, 5 L, 10 L, and 20 L. Configuration drawings of these biocontainer assemblies can be found online in the TepoFlex® Biocontainers Standards Guide.
Made of Meissner's polyethylene-based TepoFlex® film, the end-ported biocontainers have
been optimized for single-use system requirements within the biopharmaceutical industry. The biocontainers offer the industry's highest combined gas and water vapor barrier properties to provide unsurpassed protection for product stored within it.
Standard end-ported TepoFlex® biocontainers are available with silicone or TPE tubing with a variety of connections, and can be specified with or without filter capsules. Meissner offers a full selection of STyLUX® and EverLUX(TM) PES membrane filter capsules, as well as SteriLUX® PVDF membrane filter capsules. Custom end-ported biocontainers optimized for end-user's requirements can also be specified.
TepoFlex® biocontainers are certified animal component free (AFC). The film is extruded, manufactured into biocontainers and packaged in an ISO Class 7 cleanroom. For more information, please visit www.meissner.com or call Meissner Filtration Products at 800-391-9458.




