Hemoglobin Analyzer performs strip-based test.
Share:
Press Release Summary:
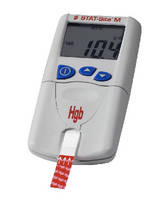
Designed for anemia determination, hand-held STAT-Site M Hgb delivers accurate hemoglobin analysis results within 30 seconds from 15 µL of finger-prick whole blood. Portable hemoglobin photometer is maintenance-free and provides step-by-step on-screen instructions. Using just one 3 V Li type battery for every 1,000 tests and with operating temperature range of 16–35°C, analyzer is suitable for field use.
Original Press Release:
EKF Launches Rapid Strip-Based Test for Hemoglobin at ISBT
Quick and easy determination of anemia using STAT-Site M Hgb
The EKF Diagnostics STAT-Site M Hgb hand-held hemoglobin analyzer
Cardiff, UK – EKF Diagnostics, the point-of-care diagnostics business, has announced the global launch of a new strip-based test for quick and easy hemoglobin analysis for anemia determination. STAT-Site M Hgb, a hand-held analyzer that combines low cost per test with portability, will be introduced at ISBT (International Society of Blood Transfusion), Amsterdam, 2-5 June 2013, Stand 128.
STAT-Site M Hgb is a pocket-sized device that reliably delivers accurate hemoglobin analysis results within 30 seconds from 15 µL of finger-prick whole blood. This hemoglobin photometer is maintenance-free, and whilst guarantees optimum performance and reliability. Intuitive, step by step on-screen instructions minimise training and ensures ease of use.
Due to its durability, portability and user-friendliness, STAT-Site M Hgb is especially suited to blood screening programmes in developing world markets, but it also has a niche in settings where operators are mobile. Using just one 3V Li type battery for every 1,000 tests and with an operating temperature range of 16° to 35°C, the analyzer is ideal for field use.
At ISBT, EKF will also be demonstrating its Hemo Control (sold as HemoPoint H2 in USA and Asia) hemoglobin analyzer designed to provide quantitative, lab quality results (imprecision < 2%) within 25 seconds for blood banks, hospitals and clinics. From one simple test this portable and robust analyzer delivers results for both hemoglobin and hematocrit and, new for 2013, includes Hemo Control Light pre-loaded data management software. The software enables users to connect directly to hospital and laboratory information systems.
Hemo Control uses unique patented microcuvette technology – the NXT microcuvette. This enables precise and easy blood collection and significantly reduces the risk of air bubbles appearing in blood samples. By minimising occurrences of air bubbles in a sample, the NXT microcuvette ensures less waste and therefore reduces cost.
For more information on EKF Diagnostics’ range of anemia screening and hemoglobin analyzers, visit www.ekfdiagnostics.com.
About EKF Diagnostics
www.ekfdiagnostics.com
EKF Diagnostics Holdings plc specialises in the development, production and worldwide distribution of point-of-care blood analyzers for use in the detection and management of diabetes, anemia, lactate and kidney related diseases. Its new Molecular division focuses on molecular and companion diagnostics.
Point-of-care diagnostics: EKF Diagnostics’ expertise covers the entire in vitro diagnostics chain, from fermentation and enzyme production, to liquid reagent manufacture, design and building of world-class diagnostic devices, and distribution of rapid test kits for infectious diseases and pregnancy. The EKF analyzer range is used widely in GP surgeries, pharmacies, blood banks, sports clinics, hospitals and laboratories for glucose, lactate, hemoglobin, hematocrit and HbA1c measurement.
Companion Diagnostics: In March 2013 EKF set up a new division to focus on molecular and companion diagnostics - EKF Molecular Diagnostics develops technologies for cancer gene detection. Through its acquisition of UK-based 360 Genomics and by offering innovative products with the potential to change current DNA extraction and detection practices, EKF is addressing the fast growing companion diagnostics market.
EKF Diagnostics products are sold in more than 100 countries around the globe. EKF Diagnostics’ strengths lie in its multi-national research and manufacturing facilities, teams of experienced analysts and engineers in Germany, Ireland, USA and the UK, and a board led by some of world’s foremost authorities in medical diagnostics.




